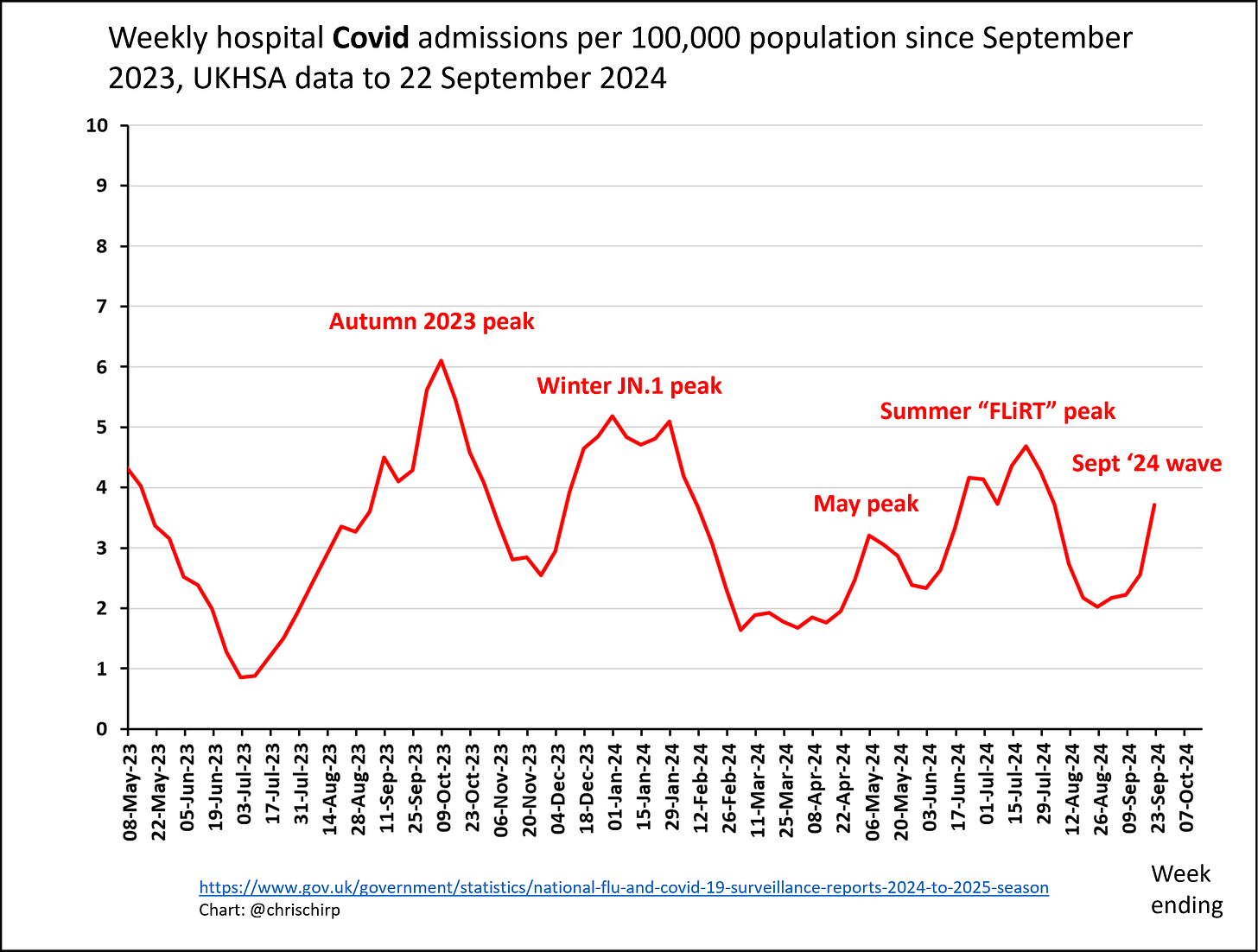
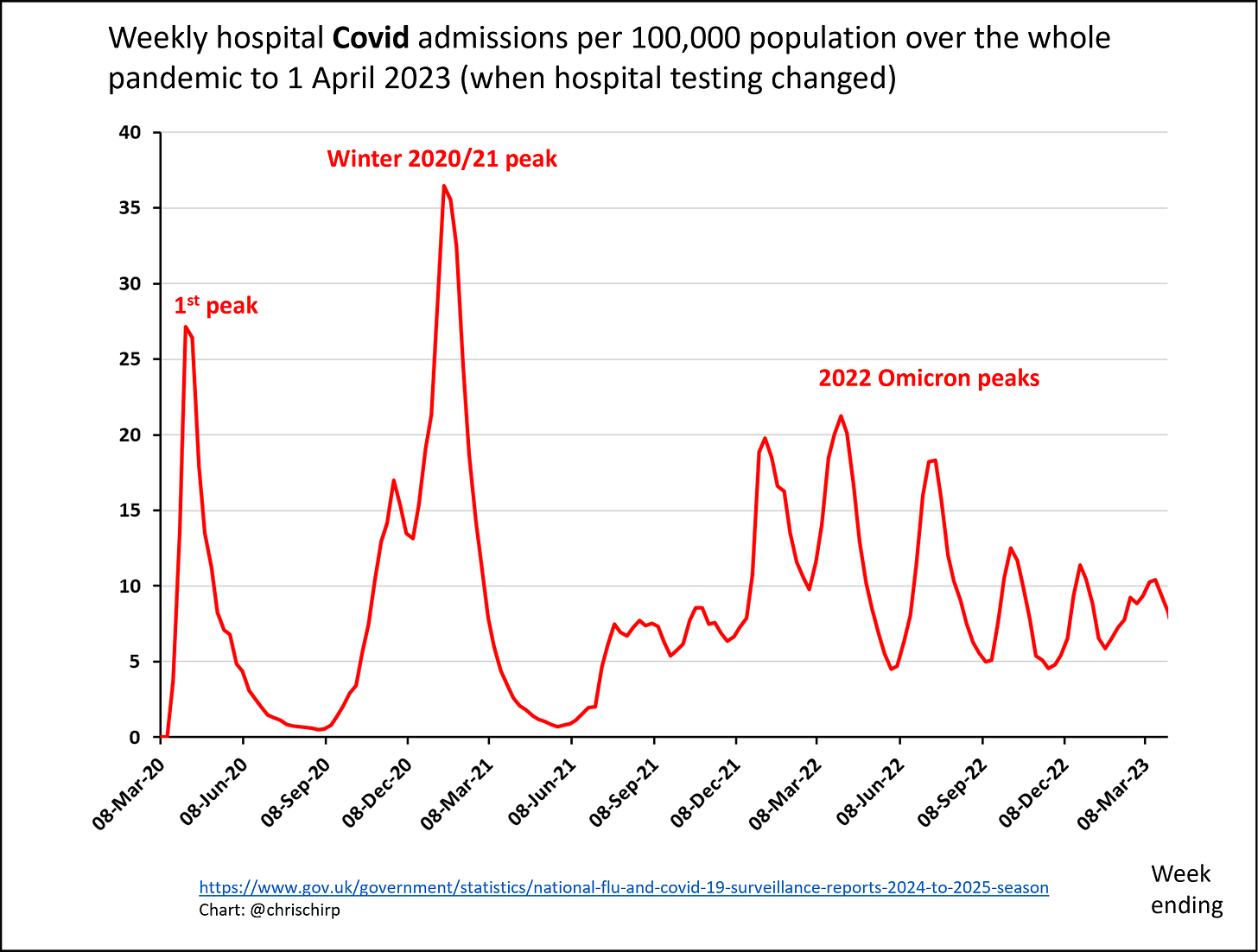
Neither vaccinations nor immunity from infections seem to thwart SARS-CoV-2 for long. The frequency of new infections within a few months of a previous bout or a shot is one of COVID-19’s most vexing puzzles. Now, scientists have learned that a little-known type of immune cell in the bone marrow may play a major role in this failure.
The study, which appeared last month in Nature Medicine, found that people who received repeated doses of vaccine, and in some cases also became infected with SARS-CoV-2, largely failed to make special antibody-producing cells called long-lived plasma cells (LLPCs). “That’s really, really interesting,” says Mark Slifka, an immunologist at the Oregon Health & Science University who was not involved with the work. The study authors say their finding may indicate a way to make better COVID-19 vaccines: by altering how they present the spike surface protein of SARS-CoV-2 to a person’s immune cells.
Durability is an age-old bugaboo of vaccine designers. Some vaccines, particularly ones made from weakened versions of viruses, can protect people for decades, even life. Yet others lose effectiveness within months. “We really haven’t overcome this challenge,” says Akiko Iwasaki, a Yale University immunologist who is developing a nasal COVID-19 vaccine she hopes can be given often enough to get around the durability problem.
Just how long a shot can protect against SARS-CoV-2 is hard to assess because variants of the virus, able to evade existing immunity, frequently emerge. And new infections muddle attempts to assess vaccine durability because they provide a “boost” that keeps immunity from waning. Multiple immune actors also provide protection, including antibodies, T cells, and natural killer cells.
To get a clearer picture, the new study examined LLPCs, which are responsible for durable immunity to some other viruses. These cells, the offspring of B cells, primarily reside in the bone marrow. For some viruses, vaccination or infection generate LLPCs that can survive for decades, steadily producing “neutralizing antibodies” that can thwart new infections.
But not so with SARS-CoV-2, the new work indicates. Emory University immunologists Frances Eun-Hyung Lee, Doan Nguyen, and their colleagues enrolled 19 people who agreed to have their marrow aspirated, a procedure that carries little risk but can be painful because it means piercing bone. All had received between two to five doses of messenger RNA (mRNA) COVID-19 vaccines—which code for SARS-CoV-2’s spike—during the preceding 3 years. Five reported having had COVID-19, as well. The study subjects had also been vaccinated recently against influenza and had booster shots for tetanus, a bacterial disease.
Lee and her colleagues found that nearly all participants had LLPCs in their bone marrow that secreted antibodies against tetanus and flu. But only one-third had plasma cells generating the same defense against SARS-CoV-2. Even in those subjects, just 0.1% of the antibodies generated by their LLPCs were specific for SARS-CoV-2, an order of magnitude less than for tetanus and flu. “The paper is very informative,” Iwasaki says.
An earlier study of bone marrow from 20 people who had been infected with SARS-CoV-2 but never vaccinated against it also found that they were “deficient” in LLPCs specific to SARS-CoV-2 compared with those for tetanus. The new results “were really consistent with what we found,” says Mohammad Sajadi of the University of Maryland School of Medicine, whose team reported the data in the 25 July issue of The Journal of Infectious Diseases. “The big question is why?”
SARS-CoV-2’s surface features may offer an answer, Lee and her co-authors say. LLPCs emerge after “naïve” B cells encounter a virus or a piece of it, such as the spike protein. As B cells mature, they make more refined antibodies that better bind to the invader. After the initial infection, memory B cells continue to patrol the blood and a subset differentiates into plasma cells. Some of those cells migrate to the bone marrow, which provides safe haven for their long-term antibody production.
B cells carry Y-shaped receptors that attach to viral surface proteins when they identify a pathogen. If both branches of the Y bind to the same pathogen proteins, they trigger a phenomenon called “cross-linking,” which spurs B cells to transform into LLPCs. But electron microscopy of SARS-CoV-2 shows its spikes are about 25 nanometers apart, too distant for a single B cell receptor to readily bind to two at once.
Spike doesn’t just appear on the virus itself; it also protrudes from infected cells and cells stimulated by mRNA vaccines. Electron micrographs don’t show the proteins and their spacing, but immunologists suspect the SARS-CoV-2 molecules are widely spaced on these cells, as well. As a result, Lee and her co-authors suggest, B cells don’t become cross-linked, and LLPCs don’t develop.
Other kinds of vaccines might present spike more effectively. Slifka points to an approved vaccine against human papillomavirus, which consists of a “viruslike particle” (VLP) made from surface proteins of that pathogen. Those proteins self-assemble into something that resembles a soccer ball. “That’s a very rigid structure with great spacing and it induces incredibly durable antibody responses,” Slifka says.
Martin Bachmann, an immunologist at the University of Bern, has argued that VLPs for SAR-CoV-2 could space spike molecules more closely—about 5 nanometers apart—than the virus itself. “I am personally convinced that viruslike particles are the best platform,” says Bachmann, who published his proposal in a 2021 npj Vaccines paper.
Given the dominance of current shots, bringing a new one to market won’t be easy. Indeed, Medicago made a spike-based VLP vaccine for COVID-19 that regulators in Canada authorized for use in February 2022, but the company stopped making it a year later because it lacked a market and went out of business.
The Novavax COVID-19 vaccine approved in the United States and some other countries uses insect cells to produce spikes that link together and form “rosettes,” which might offer tighter spacing of the protein and therefore durability benefits, but Bachmann doubts the rosettes work as well as VLPs. “Such poorly organized structures are clearly inferior to highly organized surfaces,” he says. Lee would like to study the bone marrow of Novavax recipients for the long-lived plasma cells, “but there weren’t a large number, and it’s very hard to get patients to donate marrow,” she says.
Other COVID-19 vaccines in development use nanoparticles that display tightly spaced portions of spike. Neil King, a University of Washington biochemist whose team has developed one such COVID-19 vaccine now in human trials, says they do not have data on LLPCs or durability. “Spacing definitely matters, but it’s very difficult to set up controlled experiments,” King says.
Structural biologist Pamela Bjorkman at the California Institute of Technology, who has a similar nanoparticle COVID-19 vaccine in development, is more skeptical that spacing has a significant impact on vaccine’s durability. Influenza virus has tightly spaced surface proteins, she notes, and infection with it doesn’t lead to durable immunity.
Nguyen, however, thinks his team’s sobering findings require follow-up. “The bad news is the failure of SARS-CoV-2 mRNA vaccines themselves—with or without natural infections—to induce LLPCs in the bone marrow,” he says. “The good news is this failure itself provides a research opportunity to find a way to change the fate of short-lived vaccines.”